Based within BHP’s Institute of Translational Medicine, MD-TEC is a custom-built facility designed to support medical technologies and life science businesses across the region. The team helps take products from bench to market by providing expertise and guidance on:
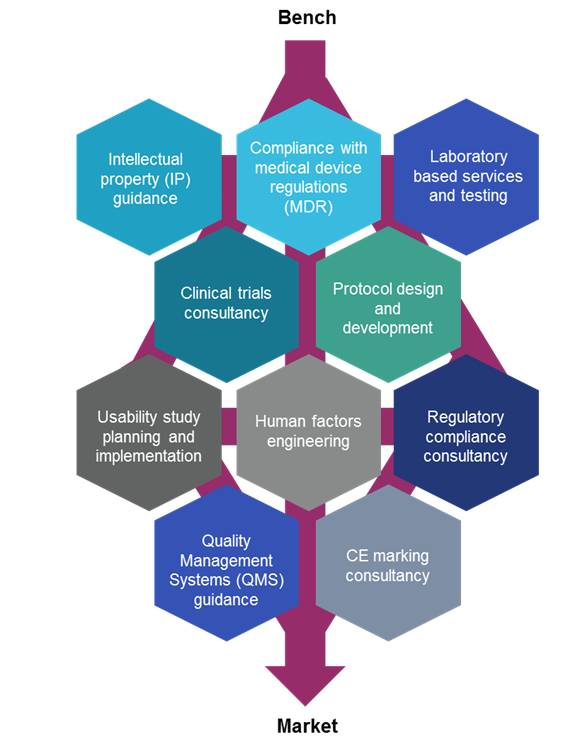
Working within realistic simulation suites and advanced laboratory facilities, MD-TEC has a broad range of expertise covering:
The team has experience in developing biomaterials from concept to finished products for clinical trials (especially those across the drug-device boundary). Key to this process is early consideration of regulations, ability to manufacture at scale and appropriate resourcing of projects.
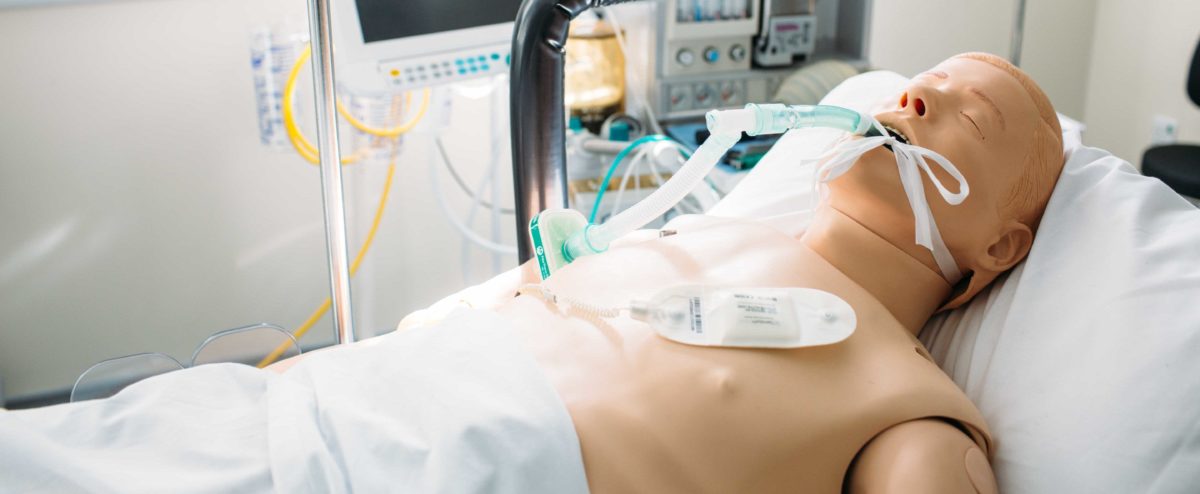
Human factors engineering is focused on the user (ergonomics) and the use (usability) of medical devices. It is usually overlooked that user error is everyone’s responsibility and in order to design a successful medical device, one must meet the requirements of the manufacturer, regulatory bodies, healthcare provider, but also of the end user. By performing usability studies at MD-TEC, the manufacturer can assure medical device safety and suitability for lay users, healthcare professionals or patients.
The Medical Device Regulations (MDR) entered into force in May 2017. Manufacturers need to demonstrate that their medical devices meet the stated requirements by carrying out a conformity assessment. The Clinical Trials Consultancy will enable you to determine if a Clinical Investigation (trial) is necessary for your medical device to pass this assessment and, if it is, support you in undertaking the trial.
There are multiple aspects involved in the process of CE marking a medical device for commercialisation across the European Economical Area (EEA). An ISO 13485 Quality Management System (QMS) for medical products and processes can help manufacturers demonstrate that they have the required systems in place before proceeding with creating a technical file for a CE mark application. MD-TEC can assist SMEs with all aspects of commercialisation.